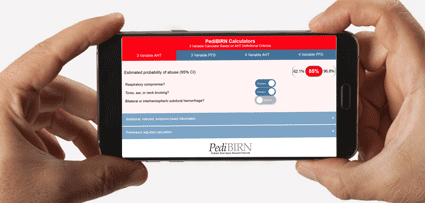
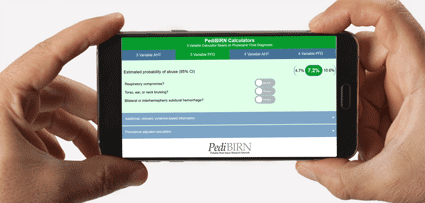
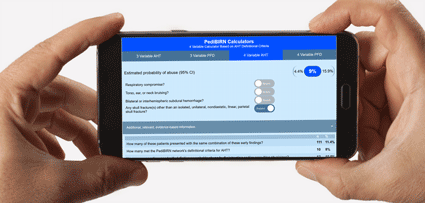
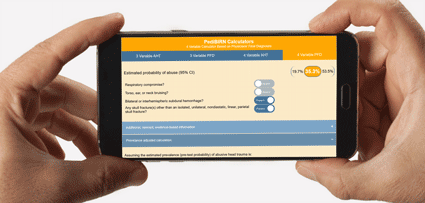
- 3 Variable Calculator Applying AHT Definitional Criteria
- Estimated probability of abuse based on:
1. Respiratory Compromise
2. Torso, ear, or neck bruising
3. Bilateral or interhemispheric subdural hemorrhage
Data Access
Access to the aggregate PediBIRN dataset is hereby granted to medical professionals seeking to conduct secondary analyses designed to inform optimal medical practice.
 Overview
Overview
This aggregate PediBIRN data set contains uniform, prospective, cross-sectional, clinical, historical, and radiological data regarding 973 acutely symptomatic patients under 3 years of age with CT- or MRI-confirmed closed head injuries hospitalized for intensive care at one of 18 academic medical centers between February 2011 and March 2021. Victims of motor vehicle collisions and patients with pre-existing brain abnormalities were excluded. The data were captured over the course of three, sequential, multicenter studies conducted to derive, validate, and implement an evidence-based screening tool for pediatric abusive head trauma. Detailed descriptions of the methods used in the parent PediBIRN studies can be found in the network's primary references.
Primary References
- Hymel KP, Willson DF, Boos SC, Pullin DA, Homa K, Lorenz DJ, Herman BE, Graf JM, Isaac R, Narang SK and Armijo-Garcia V, for the Pediatric Brain Injury Research Network (PediBIRN) Investigators. Derivation of a clinical prediction rule for pediatric abusive head trauma. Pediatr Crit Care Med 2013;14:210-220
- Hymel KP, Armijo-Garcia V, Foster R, Frazier TN, Stoiko M, Christie LM, Harper MS, Weeks K, Carroll CL, Hyden P, Sirotnak A, Truemper E, Ornstein AE and Wang M, for the Pediatric Brain Injury Research Network (PediBIRN) Investigators. Validation of a clinical prediction rule for pediatric abusive head trauma. Pediatrics 2014; 134(6):e1537-e1544
- Hymel KP, Armijo-Garcia V, Musick M, Marinello M, Herman BE, Weeks K, Haney SB, Frazier TN, Carroll CL, Kissoon NN, Isaac R, Foster R, Campbell KA, Tieves KS, Livingston N, Bucher A, Woosley MC, Escamilla-Padilla D, Jaimon N, Kustka L, Wang M, Chinchilli VM, Dias MS, and Noll J, for the Pediatric Brain Injury Research Network (PediBIRN) Investigators. A cluster randomized trial to reduce missed abusive head trauma in pediatric intensive care settings. J Pediatr 2021;236:260-268,e3.
Data Forms
The data are best interpreted in the context of the relevant data forms, provided as a separate WORD document. Across all three multicenter studies, patient-specific data were captured in their entirety as represented on data forms 2, 4, and 5. Site- and investigator-specific data captured on data forms 1 and 3 (and in section 1 of data forms 2, 4, and 5) have been excluded. The data forms include directions for PediBIRN clinician investigators, a priori definitional criteria, and embedded branching logic. Recognition and understanding of the embedded branching logic will facilitate understanding of blank data fields in the Data spreadsheet.
Data Fields
The dataset contains primary data only--that is, the data captured directly by PediBIRN investigators over the course of the network's three multicenter studies. Derived, calculated, and/or inferred data fields--based on the primary data and used in PediBIRN publications--are not included here. The data field labels that appear in column headings in the Data spreadsheet correspond to specific data fields in the relevant data form. For example, label Q2.2.1 in column A refers to form 2, section 2, question 1. For most data fields, entries of 0 conveyed a response of false, no or probably not, negative, or not selected; and entries of 1 conveyed a response of true, yes or probably, positive, or selected. For other data fields, entries of 0, 1, 2, 3, etc. reflected the corresponding 1st, 2nd, 3rd, etc. answer option listed within the specific data field in the data form (e.g., see Q5.2.1). Some data fields (e.g., Q5.5.1) were captured only in the network's third multicenter study.
Website
Information regarding the PediBIRN network's goals, screening tools, probability calculators, and publications can be found online at www.pedibirn.com. This Dataset Documentation section will be included with the data you receive, so you can refer to it there going forward.
Disclaimers
This dataset has been de-identified in accordance with HIPAA Privacy Rule's Safe Harbor standard, which includes: (1) Removal or generalization of all 18 identifiers specified by 45 CFR §164.514(b), (2) Reporting of age as ranges, and (3) Deletion of free text fields containing highly patient-specific clinical information.
The dataset is considered not to contain Protected Health Information (PHI) under HIPAA and therefore does not constitute human subjects research, in accordance with 45 CFR 46.102(e)(4) and guidance from the Office for Human Research Protections (OHRP). This dataset is provided as-is, without any representations or warranties of any kind, expressed or implied. Fourteen patients with substantive data discrepancies were excluded from the original capture of 987, resulting in a final dataset of N=973. The dataset is final and will not be updated. PediBIRN investigators are not responsible for loss or damage arising from the use of this data.
Users of this dataset must agree to:
- Indemnify PediBIRN and its investigators against any claims, liabilities, or expenses (including legal fees) arising from the use or misuse of the data;
- Use the data only for research, education, or public health purposes;
- Not attempt to re-identify any individual, nor combine this dataset with other data sources in a manner that could permit re-identification;
- Not redistribute, re-host, sublicense, or share the dataset with any third party;
- Not merge or link the dataset with external data sources in a manner that increases the risk of identification;
- Not use the dataset to develop, train, fine-tune, validate, or deploy any commercial AI/ML, predictive, or diagnostic system without prior written authorization;
- Assume sole responsibility for determining whether their proposed use of PediBIRN data requires IRB review or other institutional oversight; and
- Cite the three primary references in any publications, presentations, or derivative works.
The use of PediBIRN data is not a substitute for the application of professional medical judgment and the appropriate standard of care. Clinical interpretations or decisions should not be based on this data without confirmation from a licensed medical professional.
The network’s director reserves the right to revoke access to this dataset at any time for any reason. Upon revocation, recipients must cease use and destroy all copies of the dataset (including backups and derivatives) and certify destruction upon request.
Disclaimer | Linking Policy | Acceptable Usage Policy | Terms & Conditions (Privacy Policy)
(c) 2021 PediBIRN. All rights reserved. Website and programming by www.WebForMDs.com